
What is Repetitive Transcranial Magnetic Stimulation (rTMS)? -
- Repetitive Transcranial Magnetic Stimulation (rTMS) is a non-invasive medical treatment
- It aims to pass an electric current through the brain using pulsating magnetic fields generated outside the brain, near the scalp. This pulsating magnetic field is what creates the electric current. The locally applied electric current is what brings about the therapeutic effects for the treatment of depression, obsessive-compulsive disorder and other neuropsychiatric disorders.
- rTMS is an approved evidence-based treatment option by the American Psychiatric Association (APA), the Canadian Network for Mood and Anxiety Treatments (CNMAT), the Maudsley guidelines, the NICE (National Institute for Health and Care Excellence) of the United Kingdom, and the World Federation of Societies of Biological Psychiatry.
- In 2008, it received approval from the U.S. Food and Drug Administration (FDA). as a treatment option for patients suffering from treatment-resistant major depressive disorder and has been adopted into clinical practice in a growing number of countries worldwide such as the European Union, United Kingdom, Israel, Australia, New Zealand, USA and Canada.
Is rTMS treatment suitable for everyone? -
Absolute contraindications for TMS are any medical implant, such as a cochlear implant, a ferromagnetic foreign body in the brain within 10cm of the coil
Related contraindications include:
- History of epilepsy
- Implantable cardiac defibrillator
- Unexplained syncopal episode
- High GABAergic load
- There are no known risks in pregnancy and there are no additional risks in adolescents under 18 years of age.

TMS Safety and Side Effects:
Transcranial Magnetic Stimulation is a safe medical treatment with global safety protocols that are frequently updated. The International Federation of Clinical Neurophysiology revised its guidelines in 2020.
A common side effect (more than 1 in 100) is a transient headache or neck pain that has been described mainly in older protocols, and which passes easily with a minimal dose of
Paracetamol (Panadol / Depon). Also reported side effect is an acute auditory trauma as depending on the protocol TMS can produce 120-140db, which can be prevented by wearing earplugs by both the patient and the doctor who handles the TMS, according to new guidelines from the International Federation of Clinical Neuropsychology.
Rare side effects include: epileptic seizure, syncopal episode, toothache and hypomanic episode only in patients with bipolar disorder. According to recent studies, the probability of someone suffering an epileptic seizure from TMS is estimated to be less than 1 in 30,000 TMS treatments (i.e. <0.003%). If we calculate that each patient receives an average of 20-30 rTMS treatments, the total risk is less than 0.1%. In conclusion, many antidepressant medications have a much higher risk of causing a seizure than TMS, even though both are extremely rare.
Transcranial Magnetic Stimulation Indications and ICD-10 classification codes of mental and behavioral disorders from the World Health Organization (WHO): -
- Depressive episode (F32) and postpartum depression
- Recurrent depressive disorder (F33)
- Bipolar affective disorder (F31.5)
- Obsessive-compulsive disorder (F42)
- Mental and behavioral disorders due to use of cocaine (F14)
- Mental and behavioral disorders due to use of other stimulants (F15)
- Mental and behavioral disorders due to use of tobacco (F17)
- Anxiety disorders (F41)
- Migraine (G43)
The first official approval of rTMS therapy by the FDA in 2008 it was Resistant Depression, but since then the number of studies and evidence has increased exponentially and therefore more disorders are now approved (FDA or even CE).
Thus, TMS is now approved for the treatment of depression, unipolar or bipolar, with or without comorbid anxiety as well as for obsessive-compulsive disorder. In March 2020, the US Food and Drug Administration (FDA) granted a groundbreaking TMS approval for bipolar depression. In recent years, it has also received approvals for mental disorders due to cocaine and methamphetamine addiction, as well as for smoking cessation.
The following 4 indications, as well as many others, such as in the autism spectrum or in pain disorders such as fibromyalgia, do not yet have official approvals but have been applied in research for decades with promising results:
- Schizophrenia (F20), mainly for auditory hallucinations
- Substance use disorders (F10-19), mainly alcohol
- Eating disorders (F50), mainly anorexia nervosa
- Post-traumatic stress disorder (F43)
rTMS treatment process: -
- It is not a single treatment but a series of treatments.
- Each rTMS treatment lasts from 3 to 37 minutes / session.
- The frequency is usually 5 sessions/week, but it can vary
- The number of treatments also varies depending on the protocol and the patient's condition. In depression protocols, the duration of treatment is 4-6 weeks, i.e. usually 20 to 30 sessions. The treatment of obsessive-compulsive disorder with rTMS consists of 29 sessions lasting 18 minutes each. Finally, rTMS treatment for substance use, such as cocaine, consists of 34 sessions lasting 12 minutes each.
- Recently at Stanford University in the USA, colleagues studied a large sample of people with resistant depression who underwent 50 intensive sessions of rTMS in just 5 days with spectacular results and remission rates of 90%, that is, the most effective treatment available so far for resistant depression.
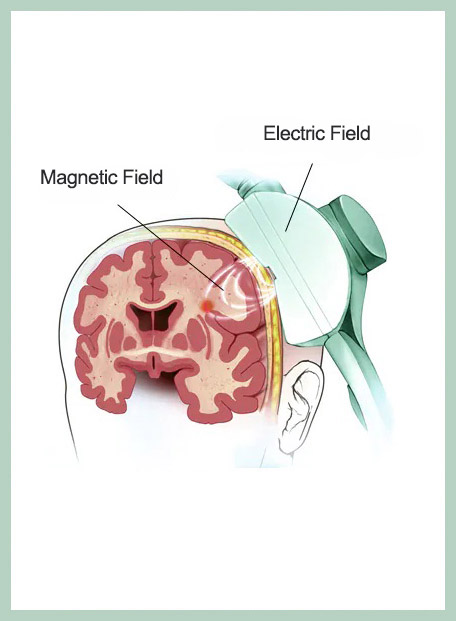
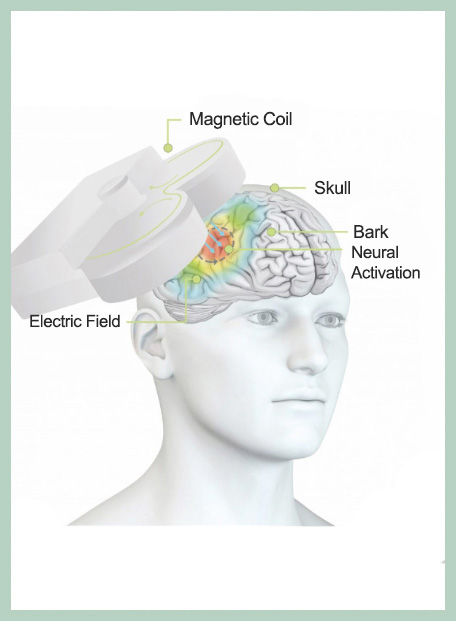
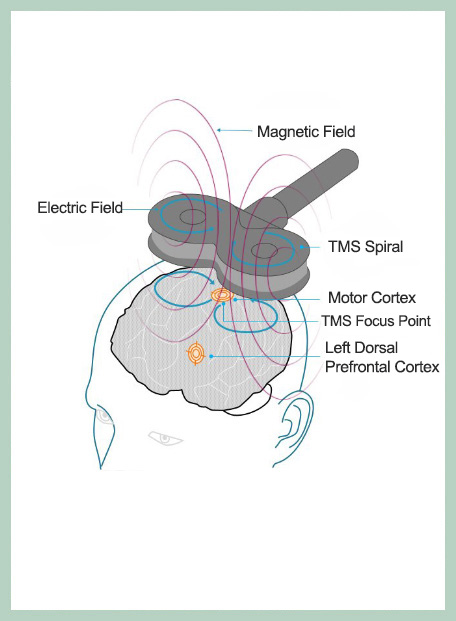
Areas targeted by rTMS: -
The rTMS coil, depending on the area of the brain it is placed in, has the corresponding therapeutic effects on the neurotransmitters it targets. More specifically, as shown in the figure below, the areas targeted by Transcranial Magnetic Stimulation therapy are
- DLPFC: Dorsal-lateral prefrontal cortex, depression and cognitive rehabilitation, 2007:O’Reardon, 2012: Fox et al
- DMPFC: Dorsal-medial prefrontal cortex, depression with prominent anhedonia, 2014:Downar et al
- TPA: Temporoparietal area, auditory hallucinations in schizophrenia, 2003:Hoffman et al
- SMA: Supplementary motor area, obsessive-compulsive disorder 2018: Lusicic et al
- Island of Reil: anorexia nervosa cessation smoking
The most studied brain region is the dorsal lateral prefrontal cortex (DLPFC).
You can see a schematic representation of the effects on the TMS network, showing how targeted focal TMS stimulation of the central executive network in the DLPFC modulates activity in other networks and anatomical parts of the brain for the treatment of depression. Thus, by stimulating a focal point in the left DLPFC, TMS induces dynamic changes in all networks of the brain. Solid arrows represent direct connectivity and dashed arrows represent indirect or sparse connectivity
When should a mental health professional refer a patient for rTMS treatment?
- In Major Depressive Disorder or Bipolar Depression: Results favor rTMS after a failed treatment with medication and also in resistant depressions.
- After starting antidepressant and/or psychotherapy
- Before Electroconvulsive Therapy (ECT)
- When there is a refusal to take medication
- When the patient has frequent side effects from antidepressants or other medications.
- In patients treated with obsessive-compulsive disorder after a failed attempt with medication or in resistant cases.
Thraki-rTMS CENTER
Services
The clinic provides, by appointment, scientifically sound psychiatric assessment, diagnosis and holistic treatment of the entire spectrum of mental disorders and psychological symptoms of adult life.





